 |
| Gel electrophoresis |
Electrophoresis is a biochemical technique used to separate charged molecules in an electric field. Gel electrophoresis is one of the most common forms of this method, used to separate DNA, proteins, enzymes, and other molecules from the cell for laboratory investigation and manipulation.
Electrophoresis is widely used to separate, visualize, or purify charged biological molecules such as deoxyribonucleic and ribonucleic acids (DNA and RNA) and proteins, including enzymes.
It is also used to estimate the size of DNA fragments and the molecular weight of proteins. Most biological molecules are electrically charged in solution; hence, when subjected to an electric field, they migrate as zones toward an electrode (a terminal source of electricity) of opposite electrical polarity.
  |
Positively charged molecules migrate to the negative electrical terminal, known as the cathode, and negatively charged molecules migrate toward the positive electrical terminal, known as the anode. The rate of migration depends on the size, shape, and charge of the molecules to be sorted as well as the strength of the electric field (voltage).
Types of Supporting Matrix
Electrophoresis is conducted in a sievelike supporting matrix, such as filter paper, cellulose acetate membrane, or, more commonly, a gel. Gels are made primarily of starch, agarose, or polyacrylamide. Starch and agarose are carbohydrates.
 |
| Types of Supporting Matrix |
Polyacrylamide is a synthetic polymer made of acrylamide and bisacrylamide. Acrylamide is a suspected human carcinogen (cancer-causing agent) and a potent neurotoxin (a compound that causes damage to the brain).
Extreme caution is therefore required when handling polyacrylamide or its components. Polyacrylamide gels have smaller pores than agarose, therefore allowing only small molecules to pass through.
Separation of Nucleic Acids
The method of choice for separating nucleic acids, typically pieces of DNA, is agarose gel electrophoresis. The DNA fragments migrate through the agarose gel at a rate that is inversely proportional to their size. In other words, smaller fragments migrate faster through the gel than larger fragments.
 |
| Separation of Nucleic Acids |
In the laboratory, an agarose gel is prepared by dissolving agarose powder in a buffer solution (a salt solution) and heating to boiling. The viscous solution formed is then cooled and poured into a casting tray.
A plastic-toothed comb is inserted in the melted agarose at the top. The agarose is allowed to solidify in the tray into a gelatinous slab and is then submerged into a buffer solution in a horizontal chamber.
The buffer functions as a conductor of electricity through the agarose gel. After the gel is submerged, the comb is carefully removed, thereby creating a row of wells in the gel slab. The wells are then loaded with a sample consisting of a mixture of DNA fragments, sucrose or glycerol, and a blue dye.
Sucrose sinks the DNA sample into the wells, while the dye marks the migration of the invisible DNA fragments through the gel. In order to establish an electric field in the chamber, a constant electric current from a power supply is generated between the electrodes at both ends of the gel.
DNA is negatively charged because of its phosphate groups; therefore, the electric current drags the DNA fragments out of the wells toward the anode through a path known as a lane. Greater voltages result in faster migration of DNA fragments through the gel. The current is switched off when the blue dye moves about three-fourths of the way.
Upon completion of electrophoresis, the separated DNA fragments are made visible by staining the gel with ethidium bromide or methylene blue. Ethidium bromide is a fluorescent dye, a potent mutagen, and possible carcinogen. The stained gel is viewed with the aid of an ultraviolet box called the transilluminator.
The separated DNA fragments appear as fluorescent orange bands. Each band corresponds to DNA fragments of equal length that have migrated to the same position in the gel. Methylene bluedye stains DNA bands blue under visible light.
   |
Separation of Proteins
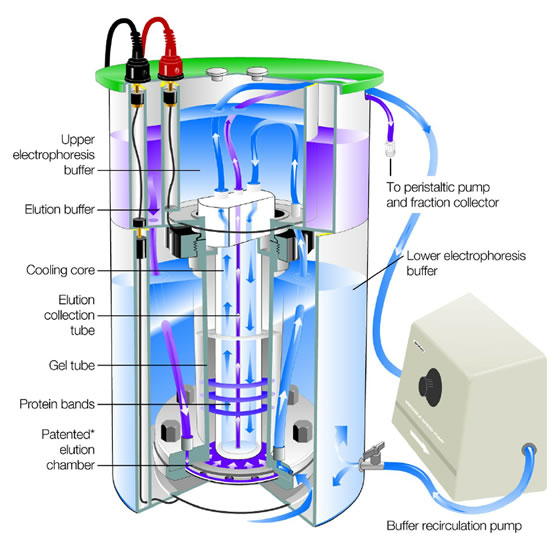
The principle behind the separation of proteins is similar to that of nucleic acids. Proteins can be separated by paper or cellulose acetate electrophoresis by simply placing a protein sample on a strip of filter paper or cellulose acetate saturated with a buffer, dipping the ends of the strip into chambers of buffer, and subject the strip to an electric field. The separation of most proteins, however, is performed in a polyacrylamide gel. The gel is cast and submerged in a vertical chamber of buffer.
Proteins can be separated on the basis of size (molecular weight) alone, net charge alone, or size and charge together. A common technique for separating proteins by size only is sodiumdodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). In this type of separation, a protein mixture is treated with the detergent sodium dodecyl sulfate.
The detergent binds and causes the proteins to dissociate into polypeptides and become negatively charged. The proteins thereafter separate into bands according to their sizes alone. Bands are then visualized by staining with silver stain or a protein dye called coomassie blue.
Proteins can be separated on the basis of charge alone, using a method called isoelectric focusing. The separation is performed in a glass tube of polyacrylamide gel in which a pH gradient has been established.
 |
| Separation of Proteins |
When a current is applied, each protein migrates until it reaches its characteristic pH (acidity or alkalinity level). At this point, the net charge on the protein becomes zero, and migration stops. The pH at which the net charge is zero is called the isoelectric focusing.
Complex mixtures of proteins of similar sizes are separated based on size and charge using the two-dimensional gel electrophoresis. In this technique, proteins are separated in two sequential steps.
First, they are separated in a tube gel by isoelectric focusing based on their charges alone. Then the proteins migrate into a gel slab and separate by SDS-PAGE, based on their size alone. The proteins are visualized as spots in the gel slab.
Applications
Every species of organism examined by researchers has revealed immense genetic variation or polymorphism (many forms), an indication of the presence of different genotypes (genetic makeup) in the population. It is however, impossible to infer the genotypes of plants simply by observing their visible characteristics or phenotypes.
In many plant science laboratories, researchers employ electrophoresis to determine the mode of reproduction of plant species, to detect genetic variation within and between plant populations, and to identify plant genotypes.
Also, researchers establish genetic relatedness in plants, that is, establish the most probable paternal parent or pollen donor within and outside a study site that sired seeds collected from a known maternal plant. To accomplish these tasks, researchers rely upon protein and DNA markers generated by gel electrophoresis.
Protein markers known as allozymes have been used extensively in a number of genetic analyses. Allozymes are electrophoretically distinct forms of an enzyme produced by different alleles (alternate forms of a gene).
An enzyme is a protein that speeds up the rate of a chemical reaction in an organism, without being consumed in the process. Allozymes catalyze the same chemical reactions but have slightly different sequences of amino acids, the building blocks of proteins.
To analyze allozymes, researchers extract enzymes from plants and separate them on starch or polyacrylamide gels. Gels made up of potato starch are most commonly used because of their low cost and ease of use.
After electrophoresis, the gel is submerged in a solution containing a dye and a substrate appropriate for the enzyme studied. The enzyme reacts with the substrate to produce a colored band on the gel.
If the gel yields one colored discrete band in a lane, then that particular plant contains just one form of the enzyme, therefore the genotype of the plant must be homozygous (having two identical alleles of a gene).
If the gel yields two colored bands, then the plant contains two forms of the enzyme and is therefore heterozygous (having two different alleles of a gene). Allozyme electrophoresis enables researchers to learn about the mode of reproduction of plants.
For example, plant populations with high numbers of heterozygotes indicate a high level of cross-pollination, that is, the transfer of pollen (plant male gamete) by wind, insects, birds, bats, or other animals from one flowering plant to another.
High numbers of homozygotes within a plant population indicate a high level of self-pollination, the transfer of pollen within a flower or between flowers of the same plant.
In many plant laboratories today, protein markers have been superseded by DNA markers, which are fragments of DNA that are distinguished from one another because of the differences in their base sequences.
To generate DNA markers, DNA is extracted from plants and cut into fragments with special enzymes known as restriction enzymes. The fragments are then separated by electrophoresis on an agarose gel and analyzed.
Some of the widely used DNA markers in many laboratories are different lengths of DNA fragments, known as restriction fragment polymorphisms (RFLPs) or short sequences of DNA bases that are repeated many times in tandem (head to tail), called variable number of tandem repeats (VNTR), a type of RFLP.
RFLP analysis distinguishes between heterozygous and homozygous genotypes. Heterozygotes yield two fragments on a gel, while homozygous genotypes yield a single fragment.
Another DNA marker, known as random amplified polymorphic DNAs (RAPDs), have become extremely popular DNA markers with plant scientists. They are DNA fragments amplified by a technique known as polymerase chain reaction (PCR) using a short primer consisting of ten nucleotides chosen randomly and then separated by size by agarose gel electrophoresis.
Polymorphisms are revealed when a DNA fragment is amplified in one plant and fails to amplify in another. On a gel, the researcher checks for the presence or absence of the marker.
Many studies involving parentage of seeds and seed pods have been done using DNA markers. The DNA profiles of seeds or seed pods are compared to maternal and paternal plants. If seeds were produced by cross-pollination, then half of the DNA bands of the seeds would be found in the maternal plant and the other half from the paternal plant.