 |
| Estrogens From Plants |
While the female hormones called estrogens are common in mammals, only a few plants contain estrogens. Others synthesize compounds which are chemically unrelated to estrogens but resemble them in their molecular size and shape. These compounds are called phytoestrogens (plant estrogens) and may, when ingested by animals or humans, have properties similar to those of mammalian estrogens.
The precursor of estrogens in plants and animals is the linear (straight-chain) triterpene known as squalene. Cyclization of squalene, via the intermediate cycloartenol in plants and via the intermediate lanosterol in animals, forms a group of very important compounds known as the steroids.
Steroids include cholesterol, mammalian sex hormones (including the estrogens and androgens), corticosteroids, insects’ molting hormones, and plant brassinosteroid hormones.
  |
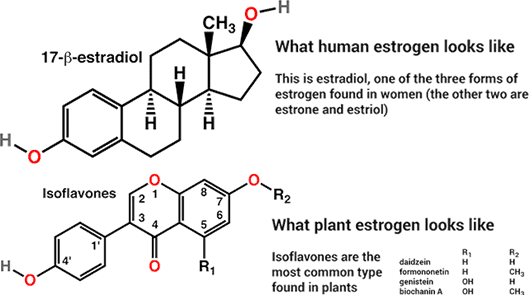
All steroids have a tetracyclic (four-ringed) structure; the rings are named A, B, C, and D. Differences in the functional groups attached to the tetracyclic skeleton, differences in the side-chain attached to ring D, and differences in the overall shape of the molecule determine a steroid’s biological activity.
Estrogens have an aromatic A ring (a ring of six carbon atoms joined by alternating single and double bonds). This constrains the junction between the A and B rings, resulting in a “flat,” or planar, molecule. This shape is essential for the potent biological activity of estrogens, and chemical modifications that alter the planar nature of an estrogen molecule reduce its biological activity.
Estrogens in Plants Versus Animals
In animals, one of the most potent estrogens is estradiol. It triggers the production of gonadotropins leading to ovulation. It is metabolized to the less active estrogens, estrone and estriol. Estrone and estriol are produced by the placentas of pregnant mammals, and both compounds accumulate in the urine during pregnancy.
In plants, estrogens are secondary metabolites. Although many thousands of secondary metabolites occur in plants, the distribution of particular secondary metabolites is often limited to just a few genera. This appears to be the case for estrogens.
 |
| the seeds of pomegranate |
Estrone has been isolated from the seeds of pomegranate and from the date palm, in which it is a component of the kernel oil. Estriol has been isolated from the pussy willow. It is not known what function these estrogens have in plants. It is possible that, like other secondary metabolites, they may function in plant defense.
Isoflavonoids as Phytoestrogens
Isoflavonoids are a type of secondary metabolite and are found almost exclusively in the legume (pea) family of plants. They are known to function in plant defense. They have been shown to deter herbivores and also to facilitate a plant’s defense response to pathogen attack. Interestingly, some isoflavonoids have chemical structures that, in overall size, shape, and polarity, resemble estrogens.
The resemblance includes the flatness, or planarity, of the molecules and the positions and orientation of oxygen atoms. Isoflavonoids that have these molecular characteristics can mimic the biological activity of estrogens and are called phytoestrogens.
   |
In terms of biosynthetic origins and chemical structure, phytoestrogens and estrogens are quite different. Phytoestrogens, being isoflavonoids, are phenolic compounds, formed from phenylalanine (an amino acid) by the shikimate pathway. In contrast, estrogens are triterpenoids, formed from acetyl coenzyme A by the isoprenoid pathway.
Isoflavonoids that are considered to be phytoestrogens exhibit only weak estrogenic activity in animals and humans. Examples of phytoestrogens are coumestrol, daidzein, and genistein. Coumestrol and daidzein are found in alfalfa (known as lucerne in Europe) and clover. Both of these plants belong to the legume family and are important forage crops for animals.
If the content of phytoestrogens in alfalfa or clover is high, the reproductive cycles of grazing animals may be adversely affected. This can pose a problem for farmers wanting to breed livestock in the normal way. For this reason, the amount of grazing in fields of alfalfa or clover has to be restricted.
Alternatively, varieties of alfalfa or clover that have been bred to contain lower levels of isoflavonoids can be grown. Unfortunately, plant varieties with lower isoflavonoid content are often more susceptible to both pathogen attack and attack by herbivorous pests.
Phytoestrogens in the Human Diet
 |
| Phytoestrogens in the Human Diet |
Dietary supplements, which are often pills, powders, or tinctures containing plant derived products, can be purchased over the counter. In the United States, the manufacture and sale of such products, classified as “dietary supplements,” is far less closely regulated and standardized than the manufacture and sale of food and drugs.
Genistein has been promoted as a possible preventive treatment or therapy for several diseases and conditions. There are claims that it reduces hot flashes associated with menopause, that it can prevent or delay the onset of osteoporosis in postmenopausal women, and that it can lower blood cholesterol levels.
In each instance the potential effectiveness of genistein would be attributable to its acting as an estrogen replacement in older women, in whom the level of estradiol is naturally low.
Genistein may also be effective in the treatment of certain breast cancers that require estrogen in order to grow. In this case it is theorized that the genistein, with weak estrogen activity, acts to reduce cancer growth by competing with the more potent estradiol for the estrogen receptor.
Some of the evidence for the role of phytoestrogens in women’s health is circumstantial. It is based, in part, on observations that women who live in countries such as Japan and China, where soy products are widely consumed, have a lower incidence of diseases such as osteoporosis and breast cancer.
Clearly, other factors, genetic and environmental, may be contributory. Health claims attributed to phytoestrogens, including genistein, need further evaluation in well-designed clinical trials before such claims can be accepted by the scientific and medical communities or relied upon by those using dietary supplements.