 |
| Photosynthetic Light Absorption |
Photosynthetic light absorption involves plants’ use of pigments to facilitate the conversion of light energy into chemical energy.
Photosynthesis occurs in green plants, algae, and certain types of bacteria. There is considerable variation among the types of pigments found in these different groups of organisms, but the basic mechanisms by which they absorb light are similar. Photosynthetic pigments are always attached to membranes within a cell.
In algae and higher plants, the photosynthetic pigments are located in the chloroplast, where photosynthesis takes place. The pigment molecules are not dispersed randomly within the chloroplast but are arrayed on the surface of the thylakoid membranes.
  |
The chloroplasts become oriented in such away as to present a large surface area to the sun or other light source, thereby maximizing the ability of the pigments to absorb light energy. In photosynthetic bacteria, the light-absorbing pigments are not organized in chloroplasts but are located on membranes that are dispersed throughout the cell.
Chlorophylls and Carotenoids
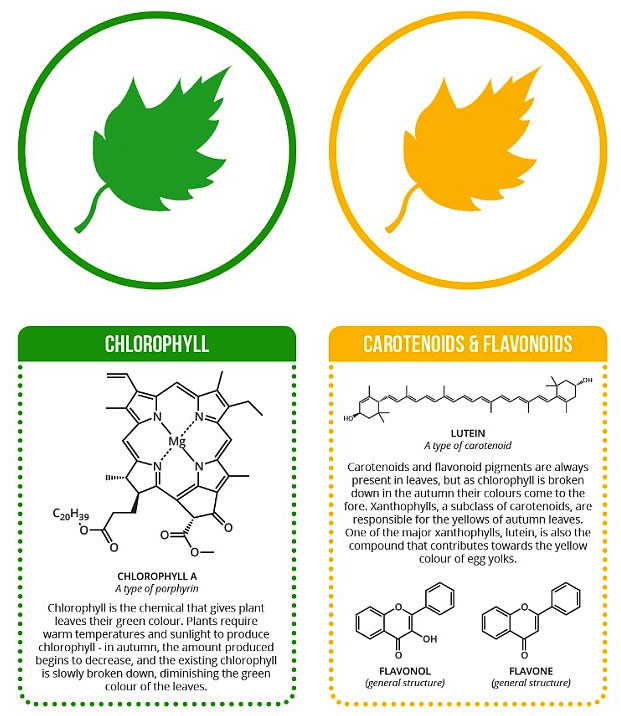
The two primary types of pigments utilized by most photosynthetic organisms are the green chlorophylls and the yellow to orange carotenoids.
There are several forms of chlorophyll that differ from one another in small details of their molecular structures. The forms are designated chlorophyll a, b, c, and so on. Chlorophyll a is found in all plants and algae, while the other forms are dispersed among various taxonomic groups.
The chlorophyll molecule consists of two parts: an elaborate ring structure that actually absorbs the light, and a long tail-like section that anchors the molecule in the membrane. Photosynthetic bacteria contain similar pigments called bacteriochlorophyll a and b.
 |
| Chlorophylls and Carotenoids |
Carotenoids, the other major group of photosynthetic pigments, also occur in various forms and are found in all types of photosynthetic plants, algae, and bacteria. The xanthophylls, which are oxygenated carotenoids, form another widespread and diverse subgroup of pigments.
Carotenoid molecules have elongated structures and, like the chlorophylls, are embedded in the photosynthetic membranes. It is a popular misconception that the change in leaf color that takes place in the fall is the result of the formation of new yellow or orange carotenoid pigments.
Actually, the carotenoids are present all the time but are masked by the presence of the green chlorophyll. In fall, the chlorophyll begins to decompose more rapidly than the carotenoids, whose yellow colors are then exposed.
Chlorophyll a, or something very similar to it, occurs almost universally in photosynthetic organisms, from bacteria to higher plants, because it is an essential component of photosynthetic reaction centers. All of the other chlorophylls and carotenoids involved in light absorption are referred to as accessory pigments.
Another kind of accessory pigment found in some groups of algae and photosynthetic bacteria are the phycobili proteins, which may impart a red or blue color to the cells in which they occur. These molecules consist of a light-absorbing portion bound to a protein.
In fact, all types of pigment molecules seem to be bound to proteins within the photosynthetic membranes. These pigment-protein associations are sometimes referred to as light-harvesting complexes, a term that accurately describes their function.
Properties of Light
To understand the functioning of photosynthetic pigments, it is necessary to consider first the physical nature of light.
Visible light is only a small portion of the electromagnetic spectrum, which ranges from very short wavelength radiation, such as X rays, to extremely long wavelength radiation, such as radio waves. The visible portion of the spectrum is intermediate in wave length and ranges from blue (at the short end) to red (at the long end).
Sunlight contains a mixture of all the visible wavelengths, which humans perceive as white light. The energy of light is inversely proportional to its wavelength; blue light has more energy than an equivalent amount of red light.
Light may be thought of as consisting of either waves or particles. For purposes of studying light absorption by pigments, it is easier to think of light as particles, referred to as photons or quanta.
When a photon is absorbed by a pigment molecule, the photon’s energy is transferred to one of the electrons of the pigment. The electron is thus said to enter an excited state and contains the energy originally associated with the photon of light.
A specific kind of pigment is not capable of absorbing all the photons it encounters. Only photons of certain energy (and therefore wavelength) can be absorbed by each pigment.
For example, chlorophyll primarily absorbs light in the blue and red wave lengths but not in the green portion of the spectrum. Consequently, the green light to which chlorophyll is exposed is either transmitted through it or reflected from it, with the result being that the pigment appears green.
The color of the pigment results from the wavelengths of light that are not absorbed. Carotenoids do not absorb light in the yellow to orange portion of the spectrum and, therefore, are seen as being that color.
The process of light absorption begins when a photon of appropriate energy strikes a chlorophyll or carotenoid molecule located on a thylakoid or other photosynthetic membrane, thus causing an electron in the pigment to be raised to an excited state.
If two pigment molecules are situated adjacent to each other in exactly the right orientation and are separated by a very small distance, it is possible for the energy of excitation to be transferred from one molecule to the next.
This transfer process (referred to as Forster resonance) enables the excitation energy tomigrate throughout the array of pigment molecules that are attached to the photosynthetic membrane.
In addition to pigment molecules, the membranes also contain a smaller number of special structures called reaction centers, which consist of special chlorophyll and protein molecules arranged in a very specific fashion.
The excitation energy migrating throughout the pigment array will eventually find its way to one of the reaction centers, and there it is utilized to form new energy-containing molecules.
All of this occurs with a large number of photons and pigment molecules simultaneously. The array of pigment molecules feeding excitation energy into the reaction centers contains both chlorophylls and carotenoids and is sometimes referred to as an antenna, to indicate its role in light absorption.
This overall process constitutes the light reactions of photosynthesis. The energy-containing molecules thus formed will then be used in the Calvin cycle to convert carbon dioxide into carbohydrates: Light energy has been converted into chemical energy.
Pigment Functions
Chlorophyll a, as well as the other chlorophylls, makes up a major portion of the antenna pigments that absorb light energy and transfer it to the reaction centers. The carotenoids, which are also part of the antenna, seem to contribute in two ways to the effectiveness of the light absorption process.
First, they increase the range of wavelengths that can be absorbed. Chlorophyll absorbs mainly in the blue and red portions of the spectrum but is not effective at absorbing other wavelengths.
Carotenoids are able to absorb some of the green light that would be unusable if chlorophyll were the only pigment present, so having a combination of different pigments makes the organism more effective at using the various wavelengths that occur in sunlight. A second function of the carotenoids has to do with their ability to protect chlorophyll from damage by intense light.
   |
Under conditions of high light intensity, chlorophyll has a tendency to decompose through a process called photooxidation. The presence of carotenoids prevents this decomposition from occurring and enables the chlorophyll to continue to function effectively at light intensities that would otherwise cause damage.
Although virtually all photosynthetic organisms utilize some form of chlorophyll in light absorption, an exception is found in the halobacteria.
These bacteria live in conditions of very high salt concentration, such as the Dead Sea or the Great Salt Lake. In these bacteria is found a purple membrane containing molecules of a pigment called bacteriorhodopsin which, remarkably, is very similar to rhodopsin, the pigment found in the visual systems of higher animals.
When bacteriorhodopsin molecules absorb light, they cause a hydrogen ion (a proton of H+) to be ejected across the cell membrane, and this leads to the formation of energy-containing molecules that the bacterium can utilize for its various metabolic requirements. Research with these unique photosynthetic organisms may also lead to a better understanding of the molecular basis and evolution of vision in animals.
