 |
| Nitrogen Cycle |
The nitrogen cycle outlines the movement of the element nitrogen from one chemical state to another as it makes its way through a series of complex physical and biological interactions.
Nitrogen (N) is one of the most dynamic elements in the earth’s biosphere; it undergoes transformations that constantly convert it between organic, inorganic, gaseous, and mineral forms.
Nitrogen is an essential element in all living things, where it is a crucial component of organic molecules such as proteins and nucleic acids. Consequently, nitrogen is in high demand in biological systems.
  |
Unfortunately, most nitrogen is not readily available to plants and animals. Although the biosphere contains 300,000 terrograms (a terrogram is a billion kilograms) of nitrogen, that amount is one hundred times less nitrogen than is in the hydrosphere (23 million terrograms) and ten thousand times less nitrogen than is in the atmosphere (about 4 billion terrograms).
Atmospheric nitrogen is almost all in the form of nitrogen gas (N2), which composes 78 percent of the atmosphere by volume. The greatest reservoir of nitrogen on the earth is the lithosphere (164 billion terrograms). Here the nitrogen is bound up in rocks, minerals, and deep ocean sediments.
Even though living things exist in a “sea” of nitrogen gas, it does them little good. The bond between the nitrogen atoms is so strong that nitrogen gas is relatively inert.
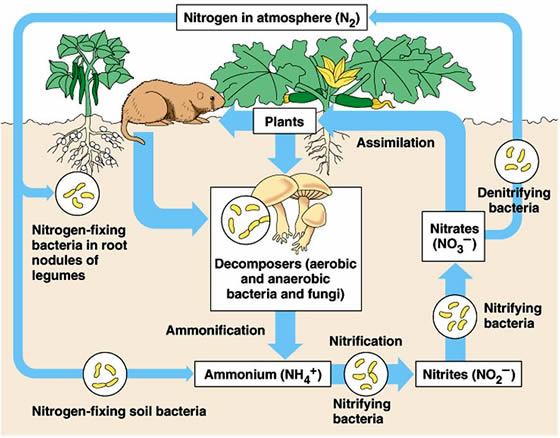
For living things to use nitrogen gas, it must first be converted to an organic or inorganic form. The nitrogen cycle is the collection of processes, most of them driven by microbial activity, that converts nitrogen gas into these usable forms and later returns nitrogen gas back to the atmosphere.
It is considered a cycle because every nitrogen atom can ultimately be converted by each process, though that conversion may take a long time. It is estimated, for example, that the average nitrogen molecule spends 625 years in the biosphere before returning to the atmosphere to complete the cycle.
Nitrogen Fixation
The first step in the nitrogen cycle is nitrogen fixation. Nitrogen fixation is the conversion, by bacteria, of nitrogen gas into ammonium (NH4+) and then organic nitrogen (proteins, nucleic acids, and other nitrogen-containing compounds).
It is estimated that biological nitrogen fixation adds about 160 billion kilograms of nitrogen to the biosphere each year. This represents about half of the nitrogen taken up by plants and animals.
The microorganisms that carry out nitrogen fixation are highly specialized. Each one carries a special enzyme complex, called nitrogenase, that allows it to carry out fixation at temperatures and pressures capable of permitting life—something industrial nitrogen fixation does not allow.
Nitrogen-fixing microbes, may be either free-living or growing in association with higher organisms such as legumes (in which case the process is called symbiotic nitrogen fixation). Symbiotic nitrogen fixation is a very important process and is one reason legumes are so highly valued as a natural resource.
Because they are able to form these symbiotic associations with nitrogen-fixing bacteria, legumes can produce seeds and leaves with more nitrogen than other plants. When they die, they return much of that nitrogen to soil, enriching it for future growth.
Mineralization and Nitrification
When plants and animals die they undergo a process called mineralization (also called ammonification). In this stage of the nitrogen cycle, the organic nitrogen in decomposing tissue is converted back into ammonium.
Some of the ammonium is taken up by plants as they grow. This process is called assimilation or uptake. Some of the ammonium is taken up by microbes in the soil. In this case the nitrogen is not available for plant growth.
If this happens, it is said that the nitrogen is immobilized. Some nitrogen is also incorporated into the clay minerals of soil. In this case it is said that the nitrogen is fixed—it is not immediately available for plant and microbial growth, but it may become available at a later date.
   |
Ammonium has another potential fate, and this step in the nitrogen cycle is nitrification. In nitrification the ammonium in soil is oxidized by bacteria (and some fungi) to nitrate (NO3–) in a two-step process.
First, ammonium is oxidized to nitrite. Next, nitrite is rapidly oxidized to nitrate. Nitrification requires oxygen, so it occurs only in well-aerated environments. The nitrate that forms during nitrification can also be taken up by plants and microbes.
However, unlike ammonium, which is a cation (positively charged ion) and readily adsorbed by soil, nitrate is an anion (negatively charged ion) and readily leaches or runs off of soil. Hence, nitrate is a serious water contaminant in areas where excessive fertilization or manure application occurs.
Denitrification
Obviously some process is responsible for returning nitrogen to the atmosphere; otherwise organic and inorganic nitrogen forms would accumulate in the environment. The process that completes the nitrogen cycle and replenishes the nitrogen gas is denitrification. Denitrification is a bacterial process that occurs in anaerobic (oxygen-limited) environments such as waterlogged soil or sediment.
Nitrate and nitrite are reduced by denitrifying bacteria, which can use these nitrogen oxides in place of oxygen for their metabolism. Wetlands are particularly important in this process because at least half of the denitrification that occurs in the biosphere occurs in wetlands.
The major product of denitrification is nitrogen gas, which returns to the atmosphere and approximately balances the amount of nitrogen gas that is biologically fixed each year.
In some cases, however, an intermediate gas, nitrous oxide (N2O), accumulates. Nitrous oxide has serious environmental consequences. Like carbon dioxide, it absorbs infrared radiation, so it contributes to global warming.
More important, when nitrous oxide rises to the stratosphere, it contributes to the catalytic destruction of the ozone layer. Besides the potential for fertilizer nitrogen to contribute to nitrate contamination of groundwater, there is the concern that some of it can bedenitrified and contribute to ozonedestruction.
The nitrogen cycle is a global cycle involving land, sea, and air. It circulates nitrogen through various forms that contribute to life on earth. When the cycle is disturbed—as when an area is deforested and nitrogen uptake into trees is stopped, or when excessive fertilization is used—nitrogen can become an environmental problem.