 |
| Autoradiography |
In 1896 Antoine-Henri Becquerel was working with rocks containing uranium ore. By chance, he put one rock sample into a dark drawer on top of a box of unexposed photographic film. When the film later was developed, it showed a clear outline of the uranium rock.
Evidently, some radiation had been emitted from the rock, penetrated through the wrapping paper, and exposed the film inside. An autoradiograph, that is, an image produced by radioactivity, was visible on the film. Autoradiography, much refined, is now a valuable technique for investigating biological processes.
Hungarian chemist Georg von Hevesy pioneered the use of radioactive tracers in biological research in the 1920’s, and two developments in the 1930’s greatly expanded their use. Firstwas the discovery of induced radiation by Frédéric Joliot-Curie and Irène Joliot-Curie, which raised the exciting possibility that artificially created radioactivity could be induced in almost any element found in nature.
  |
The second development was the invention of the cyclotron. The cyclotron beam was used to bombard various elements to produce new radioactive isotopes, including radioactive sodium, potassium, sulfur, and iron. After 1950 radioactive hydrogen and carbon also became available as tracers, allowing organic molecules such as carbohydrates and proteins to be labeled.
Macroautoradiography
Whole-body autoradiography has been widely used to trace the routes of molecules in metabolism. First, a radioactive tracer is administered to an organism by ingestion or injection. After a period of time, individual samples of tissue are removed and pressed directly against X-ray film for several days, to expose the film wherever the radioactivity has become concentrated.
The film is then developed and viewed, frequently with the aid of a microscope. This process has been used to trace the uptake of nutrients by plants from the soil into the leaves or buds. Experimentation with whole organisms is called macroautoradiography.
Microautoradiography
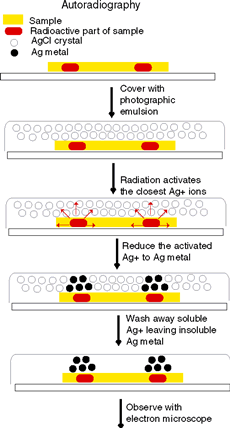
A refinement of this methodology, called microautoradiography, has been developed for studying subcellular structures, even those as small as individual strands of deoxyribonucleic acid (DNA). Much interesting information has been learned about the mechanisms of cell division and other processes in cell biology. The cells being studied are given a nutrient solution containing molecules that have been labeled, usuallywith radioactive tritium, carbon, or phosphorus.
After a period of incubation, some cells are transferred to a glass slide. The slide is dipped into a liquid photographic emulsion containing light-sensitive silver bromide, which clings to the slide in a thin layer.
The slide with the cells covered by emulsion is then placed into a light-tight box for several days to allow time for radioactive decay. The beta particles from tritium cause the photographic emulsion to become exposed.
The emulsion is then developed and fixed as any photographic negative would be. The developer washes the soluble silver bromide away and leaves behind the insoluble grains of silver, which show up as small black dots. A stain may be applied to show the outlines and structures within.
Finally, the cell is examined with a microscope. Autoradiographs typically show the black dots of exposed silver grains against a faint background of the surrounding cell structure. When higher magnification and resolution are desired, an electron microscope can be used.
In some studies, the radioactive nutrient is supplied to the cell for a short time interval, perhaps only a few minutes. This procedure is called pulse-labeling. Only those molecules that are being freshly synthesized in the cell during the "pulse" will incorporate radioactive atoms. Autoradiography will then show which cells were active.
When autoradiography is applied to chromosomes or other subcellular structures, the matter of resolution becomes very important. For high resolution to be obtained, the radioactive particles should have a short range within the photographic emulsion; the black dots of silver in the developed film should pinpoint the source of radioactive decay as precisely as possible.
Tritium works very well because it emits low-energy beta particles, which travel only a few millimeters in the emulsion, producing a well-localized image on the film. Radioactive carbon 14 emits higher-energy beta particles, so the silver grains in the film are more diffuse and the resolution is not as high.
Autoradiography and Electrophoresis
Autoradiography also has been very useful in biochemistry research when combined with the methodology of electrophoresis. Living cells are exposed to radioactively labeled amino acids, which are gradually absorbed into the proteins.
For electrophoresis, the cells are transferred to a gel to which a voltage has been applied. The protein molecules will diffuse along the gel and be sorted out by their relative molecular weights. A photographic emulsion then is placed over the gel.
Radioactivity from the proteins exposes the film, producing an image with black spots that show the distances that the different molecules drifted in the gel. The relative molecular weight of complex molecules that contain many thousands of atoms can be determined in this way.
One commercial catalog of radioactive materials lists many hundreds of organic chemicals that have been labeled with radioactive tritium. The other most common radioactive isotopes used for autoradiography are carbon 14, phosphorus 32, and sulfur 35. A large inventory of labeled chemicals has become available for the continuing use of radioactive tracers in biological research.
Applications
Autoradiography has been used in biology on the macroscopic level to study the uptake of radioactive tracers by both plant leaves and animal organs. Since the 1960’s the technique has been applied to successively smaller structures, such as individual cells, chromosomes and organelles within a cell, strands of DNA, and protein molecules. It is easier to understand the microscopic applications after first looking at a large-scale example.
In one experiment, bean plants were grown in a nutrient solution containing radioactive phosphorus. The phosphorus moved from the roots to the leaves as expected, shown by an autoradiograph of a leaf pressed against photographic film.
When the bean plant is allowed to continue growing in a nonradioactive solution, autoradiography shows that radioactive phosphorus is withdrawn from older leaves and translocated to new leaves and buds. Evidently, nutrients not only travel up from the roots but also move around the plant.
In another experiment, a solution containing phosphorus was sprayed directly onto the leaf surface and was shown to migrate away from it. Redistribution of nutrients on an even larger scale takes place in deciduous trees, where as much as 90 percent of some minerals are withdrawn from leaves before they fall.
Practical Applications
In agricultural research, the effectiveness of herbicides, insecticides, and fertilizers is studied to determine which ones can increase productivity without causing serious environmental problems. Radioactive phosphorus can be used in this regard to study plant metabolism.
The uptake of iron or zinc from the soil and their circulation in a plant can be studied to ascertain the effect of soil acidity and chemical form. Sometimes the presence or absence of other elements can inhibit translocation of an essential nutrient.
New plant growth regulators may move from one plant through the soil to a nearby untreated plant. Autoradiography is an important analytical technique for observing the route of micronutrients and discoveringwhat factors can change their mobility in a plant.
The sequence of bases in DNA molecules can be decoded by using electrophoresis combined with autoradiography, and the study of DNA sequences is crucial to research in many diverse areas of biology. Although alternatives to using autoradiography in DNA sequencing are now common, autoradiography is still a standard technique used in many other aspects of molecular biology.
